THE UK’S drug regulator has warned doctors of the ‘small risk of severe acute pancreatitis’, a potentially fatal condition, in patients taking weight‑loss and diabetes drugs linked to around two deaths per week in the UK. Meanwhile, Novo Nordisk, maker of Ozempic, faces more than 1,800 lawsuits alleging the drug has caused not only acute pancreatitis but also stomach paralysis, vision loss, kidney injury, gallbladder disease and other permanent harms. If successful, the claims could cost the company up to $2billion.
Plaintiffs argue that Novo Nordisk, which earns $25billion per year from the drugs, failed adequately to warn patients and healthcare providers about the risks, despite a mountain of evidence of adverse event reports and clinical studies.
There are four companies making GLP‑1 agonist drugs, developed to treat type 2 diabetes. These drugs activate the appetite and insulin‑regulating receptor, which increases insulin, lowers blood sugar and reduces appetite, helping users feel fuller for longer. GLP‑1s have also become popular for weight loss, boosted by celebrity users.
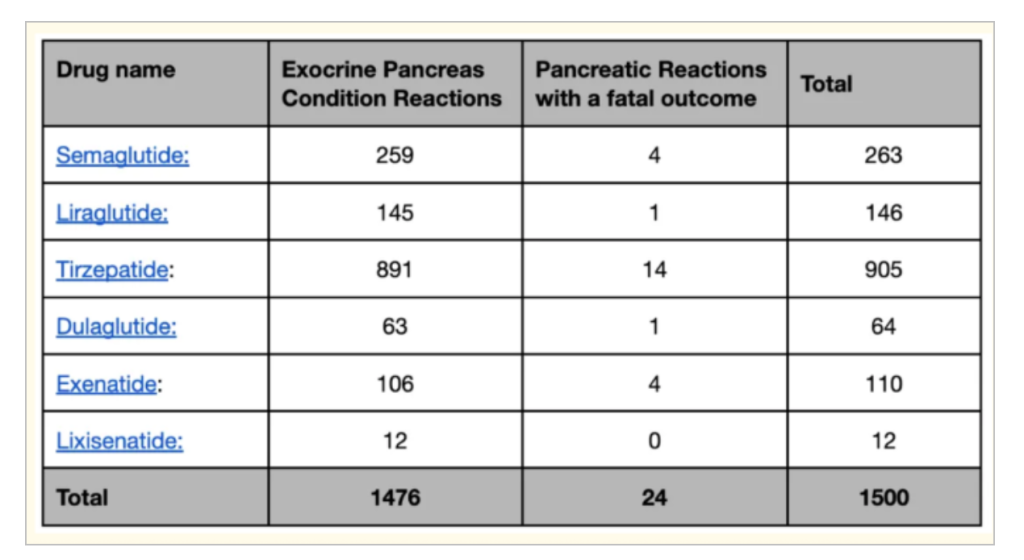
But the drugs can be lethal. In the UK, the Medicines and Healthcare products Regulatory Agency (MHRA) has received reports of 24 deaths due to pancreatic issues and 151 deaths associated with six different GLP-1 drugs.
Below is a breakdown of reports relating to pancreatic reactions and fatal outcomes.
The pancreas, a flat 10cm gland between the spine and stomach, produces digestive enzymes that help break down food in the small intestine. In acute pancreatitis, it becomes inflamed for days or weeks. Symptoms are debilitating with intense abdominal pain which is worse after eating, accompanied by nausea, vomiting, fever and rapid pulse. If the condition becomes chronic, it can cause permanent damage and becomes life-threatening if it affects other major organs, leading to shock, lung and kidney failure.
- Ozempic and Wegovy are the brand names for the active ingredient semaglutide, approved for diabetes and weight management.
- Victoza, approved for diabetes, and Saxenda, approved for weight management, contain the active ingredient liraglutide.
- Mounjaro is approved for diabetes and weight management with the active ingredient tirzepatide.
- Trulicity is approved for diabetes and includes the active ingredient dulaglutide.
- Byetta is a short-acting drug and Bydureon, long-acting approved for diabetes management contains the active ingredient exenatide.
- Lyxumia, licenced to treat diabetes, includes lixisenatide.
Novo Nordisk is the GLP-1 market leader manufacturing Ozempic, Wegovy, Victoza and Saxenda. Mounjaro and Trulicity are manufactured by Eli Lilly, who are gaining ground in the market, with $25billion annual revenue, while Byetta and Bydureon are AstraZeneca products. Lyxumia is manufactured by Sanofi.
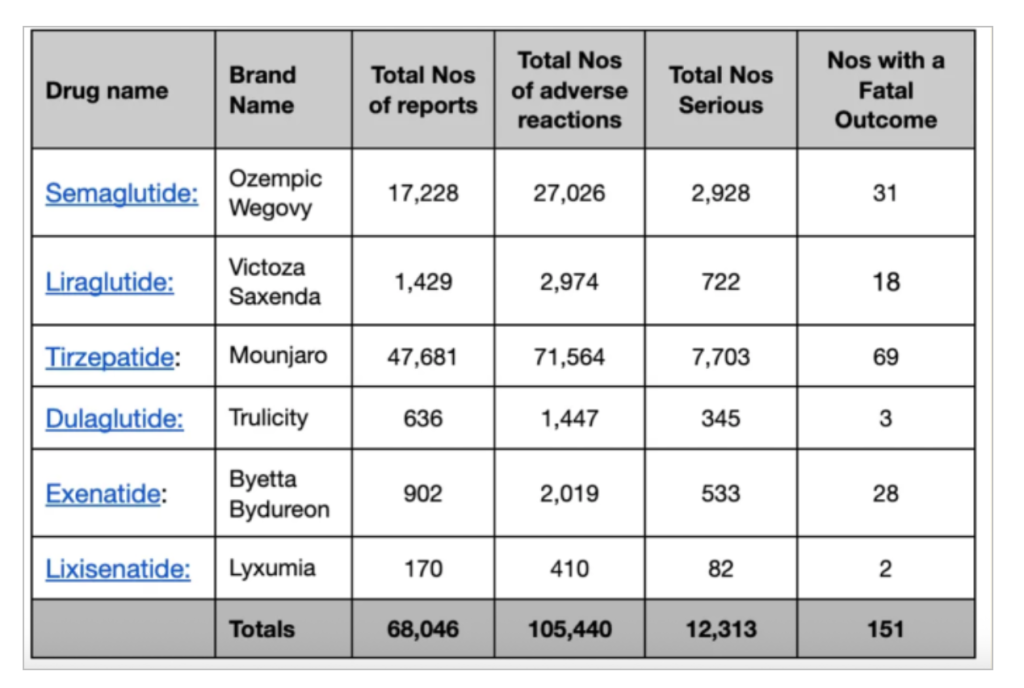
Below is a breakdown of total fatalities reported.
Tables © Trust the Evidence published by Professor Carl Heneghan and Dr Tom Jefferson. Figures reported to the Yellow Card scheme run by the Medicines and Healthcare products Regulator (MHRA)
Carl Heneghan is professor of evidence-based medicine at the University of Oxford and Tom Jefferson is senior associate tutor at the same university. They analysed the data and the reported cases of pancreatic problems linked to GLP-1 drugs and estimated that about 1.7 per cent of those reported cases were fatal. That means roughly one to two deaths for every 100 reported pancreatic reactions.
However, it’s not straightforward to compare this with the MHRA’s Yellow Card numbers. That’s because the MHRA website counts ‘reactions’, not individual reports, and one report can list more than one reaction.
Across all GLP-1 Yellow Card data there were 68,046 reports but 105,440 reactions. So, on average, each report includes about 1.5 reactions.
What that means in practice: If the MHRA shows 1,296 pancreatitis reports, that would likely translate to around 2,000 individual reaction entries, not 1,296 one-to-one cases.
A majority are using the drugs to control weight rather than diabetes or heart disease associated with obesity. In the UK approximately 1.6million adults used GLP-1 drugs to support weight loss in 2024-2025, with around 60 per cent using them exclusively for weight loss. Over a quarter (28 per cent) of British adults, 15million, are classified as obese.
In the US, 13 per cent of adults have tried drugs such as Ozempic. Figures show 43 per cent had been diagnosed with diabetes while 22 per cent took them to address obesity and weight loss. In the US, almost half, 42 per cent – 140million adults – are classified as obese.
Nurse Susan McGowan, 58, from Airdrie, Scotland, who worked at University Hospital Monklands, died in September 2024, the first recorded GLP-1 casualty after taking two low-dose injections of the weight-loss drug Mounjaro over just two weeks. She was hospitalised with severe stomach pain and sickness and her death certificate listed pancreatitis, multiple organ failure and septic shock.
The MHRA said: ‘Acute pancreatitis is a known, but infrequent side effect of taking GLP-1s. In some extremely rare cases the complications of acute pancreatitis can be particularly severe. Clinicians and patients are reminded to be alert to initial symptoms such as severe, persistent stomach pain that may radiate to the back and may be accompanied by nausea and vomiting.
‘GLP-1s are prescribed for the treatment of type 2 diabetes and, for some products, weight management, and cardiovascular risk reduction in individuals with established cardiovascular disease and a BMI of 27 kg/m2 or more. Research recently published by University College London estimates that 1.6million adults in England, Wales and Scotland used GLP-1s, such as semaglutide (Wegovy, Ozempic) and tirzepatide (Mounjaro), between early 2024 and early 2025, to lose weight.
‘While GLP-1s are generally considered safe and effective for their authorised uses, like all medications they are not without risk. Those taking GLP-1s should be aware of the symptoms of severe pancreatitis and seek urgent medical attention if they experience them.’
On January 29, Alison Cave, MHRA’s Chief Safety Officer, said: ‘Patient safety is the MHRA’s top priority and we continually monitor the safety and efficacy of all licensed medicines. For the vast majority of patients who are prescribed GLP-1s, they are safe and effective medicines which deliver significant health benefits.
‘The risk of developing these severe side effects is very small, but it is important that patients and healthcare professionals are aware and alert to the associated symptoms.
‘If you, or someone you care for, is taking GLP-1s and you notice symptoms such as severe, persistent stomach pain that may radiate to the back and may be accompanied by nausea and vomiting, we advise you speak to a healthcare professional and report it via our Yellow Card scheme.’
The Yellow Card Biobank, a collaboration between the MHRA and Genomics England, has recruited patients taking GLP-1s to investigate whether the risk of an inflamed pancreas may be influenced by an individual’s genes. It is hoped this will help to predict which patients may be most at risk of adverse reactions and prescribe the safest medicines accordingly.
Guidance on potential side effects of GLP-1s, and on the safe and effective use of GLP-1s, is available on the website.











